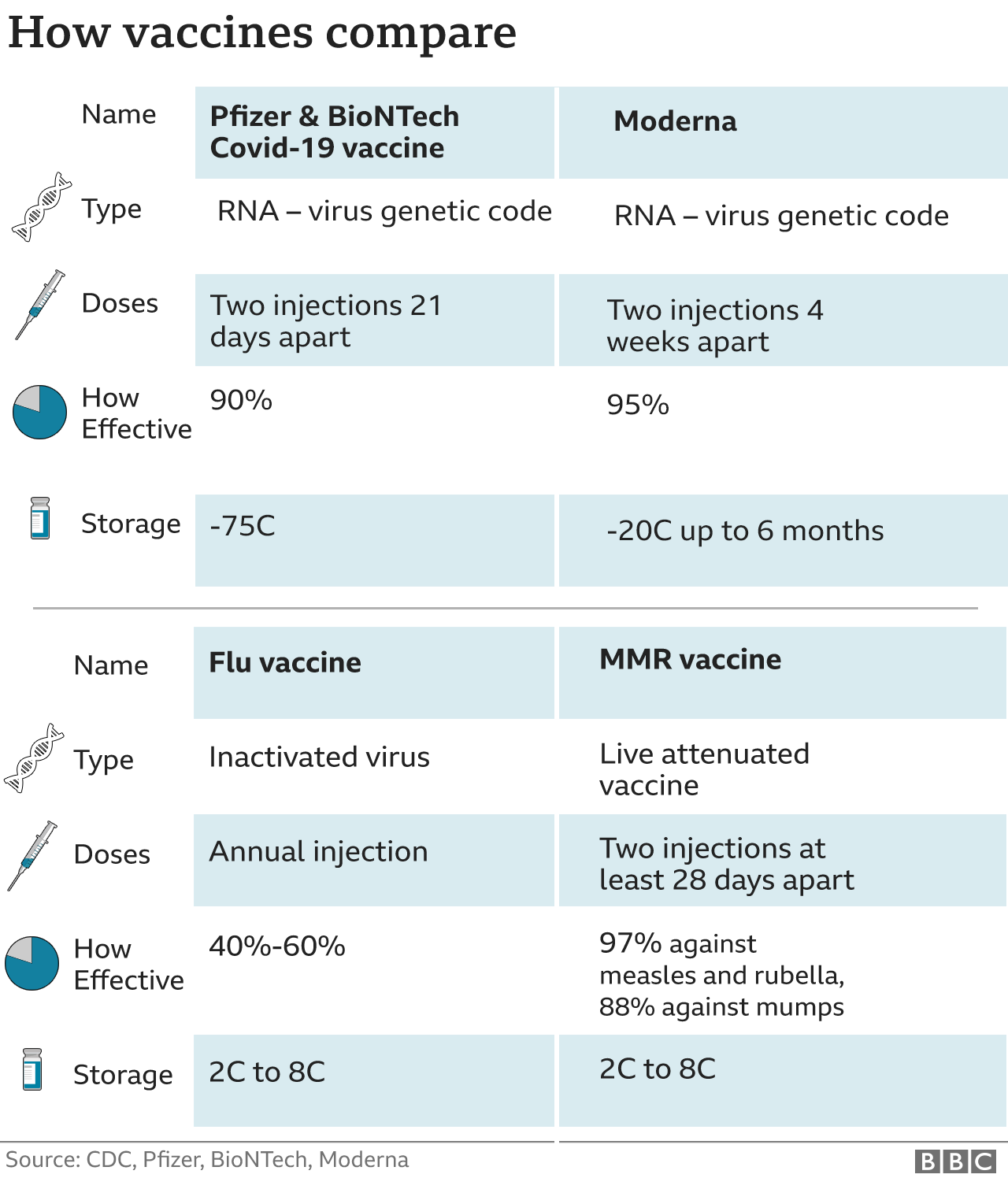
For both age groups, mRNA-1083 was compared against Spikevax booster. 
The ongoing Phase 1/2 clinical trial ( Identifier: NCT05827926) is a randomized, observer blind study evaluating the safety and immunogenicity of mRNA-1083 compared to a standard dose influenza vaccine, Fluarix, in adults 50-64 years of age and against an enhanced influenza vaccine, Fluzone HD, in adults 65-79 years of age. We are excited to move combination respiratory vaccines into Phase 3 development and look forward to partnering with public health officials to address the significant seasonal threat posed to people by these viruses." Combination vaccines offer an important opportunity to improve consumer and provider experience, increase compliance with public health recommendations, and deliver value for healthcare systems. 
"Flu and COVID-19 represent a significant seasonal burden for individuals, providers, healthcare systems and economies. "With today's positive results from our combination vaccine against flu and COVID-19, we continue to expand our Phase 3 pipeline," said Stéphane Bancel, Chief Executive Officer of Moderna. Moderna's investigational combination vaccines are designed to deliver value to individuals, providers and healthcare systems through higher compliance, easier administration and greater convenience. (NASDAQ:MRNA) today announced positive interim results from the Phase 1/2 trial of mRNA-1083, an investigational combination vaccine against influenza and COVID-19. MRNA-1083 showed strong immunogenicity against influenza and COVID-19, with an acceptable reactogenicity and safety profile, compared to licensed standalone vaccinesĬompany to begin Phase 3 trial of mRNA-1083 in adults 50 years and aboveĬAMBRIDGE, MA / ACCESSWIRE / Octo/ Moderna, Inc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
